
USP22 Promotes Therapeutic Resistance to Prostate Cancer by
Deubiquitinating Myc Leading to Increased Myc Regulated
Oncogenic Cell Transformation
Ryan Liu
a
Cedar Park High School, Cedar Park, TX 78613, U.S.A.
Keywords: USP22, Prostate Cancer, Apoptosis.
Abstract: USP22 deubiquitinating subunit serving as an oncogenic driver of prostate cancer by regulating cell
proliferation and DNA repair. USP22's negative relationship with prostate cancer is well established, but
its mechanism of action is not well defined. Here we show that USP22 is progressively overexpressed with
prostate cancer progression. USP22 controls androgen receptor signaling and promotes the expression of
various genes coregulated by androgen receptor and MYC. This reprogramming also was found to induce
transition to therapeutic resistance. Additionally, inhibiting USP22 was found to be beneficial and improved
prostate cancer prognosis. Taken together, we demonstrate the promise of USP22 as a therapeutic target for
prostate cancer.
1 INTRODUCTION
a
Prostate cancer is the second most deathly and
common type of cancer in developed countries.
More than 209, 900 American men were diagnosed
with prostate cancer in 1997 (Mazhar, 2002). The
main risk factor is age. Prostate cancer is very rare
under the age of 40, but the risk of developing
prostate cancer exponentially increases with age.
Environmental factors and race are other risk factors
as well (Prostate cancer - Symptoms and causes,
2021). This includes diet and radiation exposure.
Compared to men of other races, African Americans
have a greater chance of developing prostate cancer.
An analysis showed that exposure to radiation was
linked to cases of prostate cancer. Prostate cancer is
caused by at least eight genetic mutation events.
Most cases are due to the loss of tumor suppressive
genes. P53 and p21 are examples of genes that are
mutated in prostate cancer. Obesity is another risk
factor, as people who are obese may have a higher
risk of prostate cancer than those who have a healthy
weight.
Several treatments have been developed to help
treat prostate cancer. Combined androgen blockade
eliminates all sources of androgen. Studies have
a
https://orcid.org/0000-0002-2637-1788
shown a 2.3% increase in survival with the use of
flutamide (Mazhar, 2002).
Intermittent androgen blockade involves medical
castration and discontinuation of the GnRH agonist,
which delays the development of hormone
resistance. Androgen blockade uses finasteride and
antiandrogens (Mazhar, 2002). Finasteride is a
reductase inhibitor, meaning that it prevents
testosterone converting to dihydrotestosterone.
Dihydrotestosterone has a higher affinity for the
androgen receptor as well.
Increasing evidence has shown the deregulation
of the UPS22 deubiquitinase, a protease that cleaves
ubiquitin from proteins to cancer development.
USP22 is a promoter of the tumor phenotype by
modulating nuclear receptor, a ligand-regulated
transcription factor, and oncogenic signaling,
networks of signaling pathways that interact with
each other to control the growth and progression of a
tumor (Schrecengost, Dean, Goodwin, Schiewer,
Urban, Stanek, et al., 2013). USP22 uses
deubiquitination to modulate protein function and
promote cell proliferation. USP22 is required for
Myc function and potentiates Myc-mediated
oncogenic cell transformation in some cancers. This
enhances the expression of target genes co-regulated
by AR and Myc, a regulator proto-oncogene that
codes for transcription factors. USP22 included the
transition to therapeutic resistance (Schrecengost,
702
Liu, R.
USP22 Promotes Therapeutic Resistance to Prostate Cancer by Deubiquitinating Myc Leading to Increased Myc Regulated Oncogenic Cell Transformation.
DOI: 10.5220/0011260300003443
In Proceedings of the 4th International Conference on Biomedical Engineering and Bioinformatics (ICBEB 2022), pages 702-706
ISBN: 978-989-758-595-1
Copyright
c
2022 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved

Dean, Goodwin, Schiewer, Urban, Stanek, et al.,
2013). USP22 has been found to be a critical
effector of tumor progression that drives lethal
phenotypes. This makes the enzyme a promising
therapeutic target to treat advanced disease. The
deregulation of USP22 induces androgen-
independent AR recruitment to target gene loci and
supports cell growth without androgens. The
depletion of USP22 down-regulates AR protein
levels and evades AR activity in ADT-sensitive, a
hormone therapy that stops testosterone from being
produced, and CRPC cells, cells that express low
levels of the androgen receptor (Schrecengost, Dean,
Goodwin, Schiewer, Urban, Stanek, et al., 2013). It
was thought that USP22 may control fundamental
oncogenic signaling pathways that are implicit to
prostate cancer initiation and progression. USP22
expression promotes the activation of target genes
that are coordinated by AR and Myc. These are both
maintained without androgens and AR antagonists.
The predicted outcome is that USP22 promotes
therapeutic resistance to prostate cancer by
deubiquitinating Myc leading to increased Myc
regulated oncogenic cell transformation.
2 METHOD
Chromatin Tethering Assays
The cells were harvested from a 10 cm plate and
suspended in a 100 μL CSK buffer with protease
inhibitor (McCann, Vasilevskaya, Poudel Neupane,
Shafi, McNair, Dylgjeri, et al. 2019). After
removing μL of the suspension, the suspension
leftover was diluted with CSK buffer to a volume of
1 mL. This was incubated in ice for 20 minutes. The
chromatin-tethered assay was pelleted and extracted
in 1 mL of the CSK buffer for 10 minutes. This was
repeated and sample buffer was added. After the
sample was boiled for 5 minutes, western blot
analysis can begin.
Western Blotting
CRISPR was used to knock out an expression vector
to overexpress USP22 in LNCaP prostate cancer
cells. Ubiquitination of Myc will be measured using
western blot for Myc and looking for Ubiquitin
mediated ladders with a GAPDH western blot
loading control (Schrecengost, Dean, Goodwin,
Schiewer, Urban, Stanek, et al., 2013). Antibodies
used to detect proteins were AR-N20, USP22, Myc,
GAPDH, and β-actin. LNCaP cells were infected
with shRNA-containing lentivirus and mixed with
puromycin for 5 days (Schrecengost, Dean,
Goodwin, Schiewer, Urban, Stanek. et al., 2013).
Immunohistochemistry
TMAs were stained to search for USP22 using
USP22 polyclonal antibody (Schrecengost, Dean,
Goodwin, Schiewer, Urban, Stanek. et al., 2013). The
antibody was diluted 1:150 with detection using
LEICA polyvision+. The unstained 5 μm sections
were taken out of paraffin TMA blocks
(Schrecengost, Dean, Goodwin, Schiewer, Urban,
Stanek et al., 2013). It was incubated with the
primary antibody at room temperature for 45
minutes.
Ubiquitination
Myc deubiquitination will be measured in vitro
using ubiquitinated Myc as a substrate. Recombinant
Myc will first be ubiquitinated by either Skp2,
Fbw7, and Huwe1 and then tested for
deubiquitination by adding recombinant USP22.
Gene expression analysis
Cells were treated as listed on the packaging and
RNA was isolated using TRIzol (McCann,
Vasilevskaya, Poudel Neupane, Shafi, McNair,
Dylgjeri et al. 2019). LNCaP cells are analyzed by
qPCR using the manufacturer’s instructions.
Cell growth assays
The cell culture was made up of LNCaP and C4-2
cells. They were both maintained in IMEM and
supplemented with 5% FBS. A separate culture of
22Rv1 cells were maintained in DMEM and
supplemented with 10% FBS. Media was
supplemented with 2 mmol/L L-glutamine and 100
units/mL penicillin-streptomycin (Schrecengost,
Dean, Goodwin, Schiewer, Urban, Stanek et al.,
2013). To ensure that the cell lines were stable, the
LNCaP and C4-2 cells were transducted using
lentivirus and went through three rounds of selection
with their corresponding antibiotic. The cells were
plated at equal densities and the cell number was
measured using trypan blue exclusion with a
hemocytometer (McCann, Vasilevskaya, Poudel
Neupane, Shafi, McNair, Dylgjeri et al. 2019).
USP22 depletion
USP22 was transfected with Dharmafect and
incubated for 72 hours for siRNA-mediated
depletion. shRNA sequences that targeted USP22
were annealed, cloned, and packaged into virus for
inducible USP22 depletion (Schrecengost, Dean,
Goodwin, Schiewer, Urban, Stanek. et al., 2013).
3 RESULT
Table 1 shows USP22 and ubiquitination and Table
2 shows USP22 and apoptosis. Exhaustive lists of
possible results are listed in the two tables below.
Refer to notes.
USP22 Promotes Therapeutic Resistance to Prostate Cancer by Deubiquitinating Myc Leading to Increased Myc Regulated Oncogenic Cell
Transformation
703

Table 1: USP22 and Ubiquitination.
Possible Results PR 1A PR 2A PR 3A PR 4A PR 5A PR 6A
Knockout USP22
Increases Myc Ubiquitin
Western Blot
+ - - - - -
Overexpress USP22 in
HCC Decreases Myc
Ubiquitin Western Blo
t
+ + - + - -
USP22 Decreases
Ubiquitination Induced
by
Skp2
+ + + + - -
USP22 Decreases
Ubiquitination Induced
by
Fbw7
+ + + - + -
USP22 Decreases
Ubiquitination Induced
by
Huwe1
+ + + - + -
USP22 Decreases Myc
Ubiquitination Caused
by
SKP2
+ + + - + -
Note: “+” represents a result corresponding to the possible result and “-” represents a result contradicting
the possible result.
Table 2: USP22 and Apoptosis.
Possible Results PR 1B PR 2B
USP22 Correlates with Cell
Apoptosis Acceleration
+ -
Note: “+” represents increased level of apoptosis and “-” represents decreased level of apoptosis.
3.1 Explanation of Tables
Possible Result 1A
Knockout USP22 increases Myc ubiquitin western
blot and USP22 decreases Myc ubiquitination
caused by SKP2. This is the expected result that
aligns with the hypothesis presented.
Possible Result 2A
Knockout USP22 decreases Myc ubiquitin western
blot and USP22 decreases Myc ubiquitination
caused by SKP2.
Possible Result 3A
Knockout USP22 decreases Myc ubiquitin western
blot, overexpress USP22 in HCC increases Myc
ubiquitin western blot, and USP22 decreases Myc
ubiquitination caused by SKP2.
Possible Result 4A
Knockout USP22 decreases Myc ubiquitin western
blot, overexpress USP22 in HCC decreases Myc
ubiquitin western blot, USP22 increases
ubiquitination induced by Huwe1, and USP22
increases Myc ubiquitination caused by SKP2.
Possible Result 5A
Knockout USP22 decreases Myc ubiquitin western
blot, overexpress USP22 in HCC increases Myc
ubiquitin western blot, USP22 increases
ubiquitination induced by Skp2, and USP22
decreases Myc ubiquitination caused by SKP2.
Possible Result 6A
Knockout USP22 decreases Myc ubiquitin western
ICBEB 2022 - The International Conference on Biomedical Engineering and Bioinformatics
704
blot and USP22 increases Myc ubiquitination caused
by SKP2. This is the unexpected result that
contradicts with the hypothesis presented.
Possible Result 1B
USP22 correlates with cell killing. This is the
expected result that aligns with the hypothesis.
Possible Result 2B
USP22 does not correlate with cell killing. This is
the unexpected result that contradicts the hypothesis.
3.2
Controls
Positive control is to add a known deubiquitinase for
Myc. The positive control should be implemented to
make sure USP22 is active. The negative control is a
control treatment such as a scrambled targeting
vector and an overexpression of an irrelevant
protein.
4 DISCUSSION
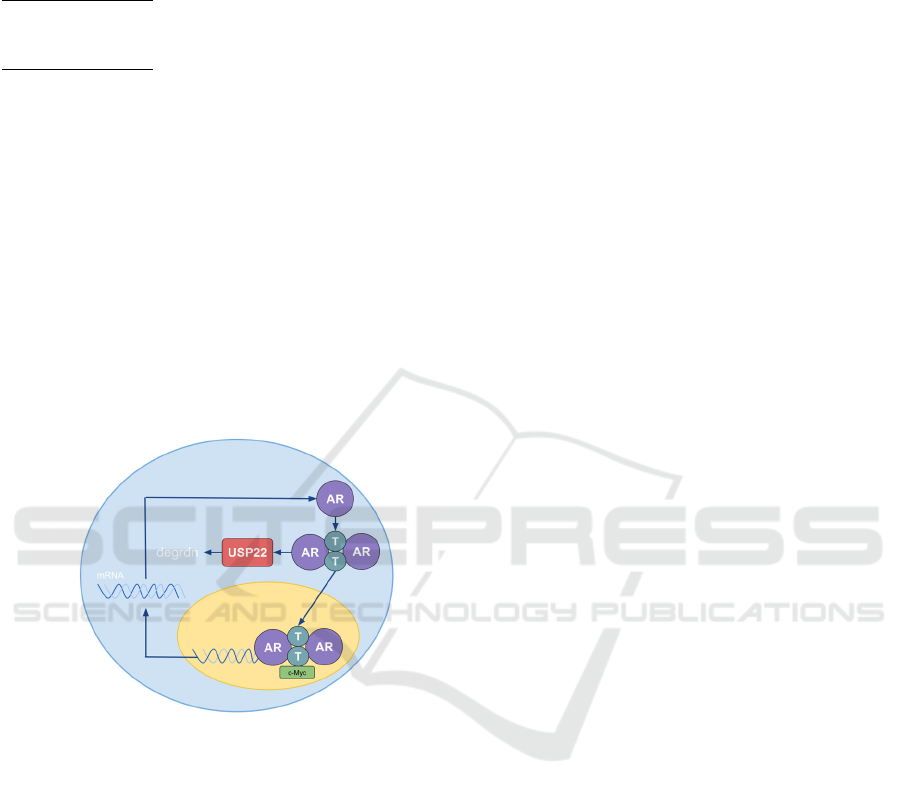
Note: AR corresponds with androgen receptor. T
corresponds with testosterone. USP22 leads to the
degradation of the intracellular hormones
(Michmerhuizen, Spratt, Pierce, Speers 2020). c-
Myc acts as the co-regulator of the transcription
inside the nucleus.
Figure 1.
Because USP22 regulates transcriptional activation
of target substrates, which in turn promotes
oncogenic phenotypes, knocking out USP22 will
decrease Myc ubiquitination rates (Chan, Lee,
Wang, Lin. 2010). Since Myc ubiquitin is
responsible for transcription regulation, decreasing
the ubiquitination rate is expected for a Skp2
deficient cell. Skp2 deficiency inhibits cancer
development, as it prevents aerobic glycolysis and
Akt activation (Chan, Lee, Wang, Lin, 2010). Since
Akt activation causes a chain reaction of Glut 1
expression, it promotes cancer development.
Inhibiting Akt activation will prevent cancer
initiation and slow its growth. Therefore, possible
result 1A is the most likely result and aligns with the
presented hypothesis. Possible Result 6A contradicts
the current understanding of the ubiquitin signaling
pathway. This result is unlikely to happen on the
LNCaP cell line. This result may occur in variations
of mutations and genetic disorders in a large group
of people. Possible result 1A is consistent with
previous studies investigating USP22’s effect on
malignant cells. Since USP22 promotes therapeutic
resistance to prostate cancer, there is an acceleration
in cell apoptosis. The relation between USP22 and
Skp2 should be investigated to find a more specific
ubiquitin pathway. Therefore, possible result 6 is
unlikely and contradicts with the presented
hypothesis.
In vitro studies, an analysis took place,
examining a range of concentrations of USP22/the
E3 ligases/Myc. This increased confidence that a
specific effect would be uncovered. It also should be
noted that the experiment was performed three times
to discern significant changes. Previous studies
report that USP22 knockdown decreased in vitro
survival of cancerous cells compared to the controls.
Doing an analyzation of the western blot of the
control and the knockout USP22 cancer cells show
that USP22 modulates the Myc FOXO1 and YAP
signaling pathways (Liu, Liu, Zhao, Zhu, Wang, Liu,
2019). Injection of knockout USP22 cancer cells
into mice generated smaller tumors than did the
control cells. This previous study overall shows that
USP22 regulates the growth and progression of
cancerous cells through the Myc dependent FOXO1
and YAP signaling pathways (Liu, Liu, Zhao, Zhu,
Wang, Liu, 2019). Previous studies have also shown
that overexpression of USP22 is associated with
enhanced angiogenesis. However, USP22 knockout
suppressed in vitro proliferation. It also impaired
non-homologous DNA damage repair capacity and
enhanced irradiation-induced apoptosis (Zhang,
Yang, Wang, Sun, Guo, Nelson 2019). USP22 is
vital for castration-resistant AR expression, cell
proliferation, and tumor growth.
As prostate cancer progresses, namely,
transitioning from hormone therapy-resistant to
castration therapy-resistant, USP22 protein
expression increases (McCann, Vasilevskaya,
Poudel Neupane, Shafi, McNair, Dylgjeri 2019).
This is because USP22 modulates AR stability and
activity. When AR is overexpressed, the prostate
cancer is able to progress to castration level of
USP22 Promotes Therapeutic Resistance to Prostate Cancer by Deubiquitinating Myc Leading to Increased Myc Regulated Oncogenic Cell
Transformation
705

androgen (Fujita, Nonomura, 2019). The prostate
cancer cells can then progress to castration-resistant
prostate cancer if AR is further amplified under
androgen deprivation circumstances. The fact that
USP22 modulates the AR plays a part in this
progression. Even after new AR-targeted therapy is
developed, castration-resistant prostate cancer will
eventually gain resistance again. This provides a
motivation to find out more about the mechanisms in
which USP22 controls AR. There are multiple
possible findings that could answer this question.
USP22 upregulation occurs in most progressions of
prostate cancer and is correlated to poor outcomes
(McCann, Vasilevskaya, Poudel Neupane, Shafi,
McNair, Dylgjeri, 2019). This USP22 alteration is
associated with pro-proliferative oncogenes,
including AR and Myc. This shows that knockout of
USP22 decreases the proliferation of cells in prostate
cancer. USP22 is enough to cause hyperproliferative
prostate cancer, which demonstrates that USP22
alone can induce pro-tumorigenic phenotypes in
previously normal tissue. All of this supports the fact
that USP22 is a critical driver of the oncogenic
phenotype in prostate cancer.
5 CONCLUSION
Although USP22 is a deubiquitinating enzyme that
has been linked to carcinogenesis, not much is
known about its function and regulation in both
cancerous and non-cancerous tissue. Further studies
investigating the specific regulation mechanism of
USP22 should be done for a more thorough
understanding of the role of USP22. These further
studies will solidify USP22’s role in cancer
development. Since not many studies of USP22 have
been done on mammals, preclinical testing on mice
should also be done to improve the therapeutic
method of a ubiquitin pathway.
USP22 is a critical effector that modulates AR
levels, AR-Myc coordination, and the progression of
prostate cancer to castration-resistant prostate cancer
(Schrecengost, Dean, Goodwin, Schiewer, Urban,
Stanek 2013). Since USP22 plays such a major role
in this progression, therapies that target USP22
would be highly beneficial to prevent the initiation
and progression of prostate cancer.
REFERENCES
Chan, C., Lee, S., Wang, J., & Lin, H. (2010). Regulation
of Skp2 Expression and Activity and Its Role in
Cancer Progression. The Scientific World JOURNAL,
10, 1001-1015. doi: 10.1100/tsw.2010.89.
Fujita, K., & Nonomura, N. (2019). Role of Androgen
Receptor in Prostate Cancer: A Review. The World
Journal Of Men's Health, 37(3), 288. doi:
10.5534/wjmh.180040.
Liu, H., Liu, N., Zhao, Y., Zhu, X., Wang, C., & Liu, Q. et
al. (2019). Oncogenic USP22 supports gastric cancer
growth and metastasis by activating c-
Myc/NAMPT/SIRT1-dependent FOXO1 and YAP
signaling. Aging, 11(21), 9643-9660. doi:
10.18632/aging.102410.
Mazhar, D. (2002). Prostate cancer. Postgraduate Medical
Journal, 78(924), 590-595. doi:
10.1136/pmj.78.924.590.
McCann, J., Vasilevskaya, I., Poudel Neupane, N., Shafi,
A., McNair, C., & Dylgjeri, E. et al. (2019). USP22
Functions as an Oncogenic Driver in Prostate Cancer
by Regulating Cell Proliferation and DNA Repair.
Cancer Research, 80(3), 430-443. doi: 10.1158/0008-
5472.can-19-1033.
Michmerhuizen, A., Spratt, D., Pierce, L., & Speers, C.
(2020). ARe we there yet? Understanding androgen
receptor signaling in breast cancer. Npj Breast Cancer,
6 (1). doi: 10.1038/s41523-020-00190-9.
Prostate cancer - Symptoms and causes. (2021).
https://www.mayoclinic.org/diseases-
conditions/prostate-cancer/symptoms-causes/syc-
20353087.
Schrecengost, R., Dean, J., Goodwin, J., Schiewer, M.,
Urban, M., & Stanek, T. et al. (2013). USP22
Regulates Oncogenic Signq2bbbbhaling Pathways to
Drive Lethal Cancer Progression. Cancer Research,
74(1), 272-286. doi: 10.1158/0008-5472.can-13-1954
Sun, T., Liu, Z., & Yang, Q. (2020). The role of
ubiquitination and deubiquitination in cancer
metabolism. Molecular Cancer, 19(1). doi:
10.1186/s12943-020-01262-x.
Zhang, K., Yang, L., Wang, J., Sun, T., Guo, Y., &
Nelson, R. et al. (2019). Ubiquitin-specific protease 22
is critical to in vivo angiogenesis, growth and
metastasis of non-small cell lung cancer. Cell
Communication And Signaling, 17(1). doi:
10.1186/s12964-019-0480-x.
ICBEB 2022 - The International Conference on Biomedical Engineering and Bioinformatics
706
